
Greta, A New ESA Rocket Engine, Successfully Completes Testing
Additive manufacturing has established itself as a fundamental technology in rocket development, especially in the production of combustion chambers, as it allows
🌸With the arrival of spring, our company has officially resumed full operation and production capacity after the Spring Festival holiday. We sincerely thank all customers for your patience and support.
To honor International Women’s Day and thank you for your trust, we are launching a limited-time special promotion:
⏰ Promotion Time
March 5 – March 31
🎯 Exclusive Discount
8% OFF Coupon for all orders placed during the valid period.
💡 Warm Tips
Place your order early to enjoy priority production and guaranteed delivery time.
This event is dedicated to celebrating the power of women worldwide and thanking you for your continued partnership.
For any inquiries, please contact our sales team.
Thank you for choosing us, and let’s move forward to a prosperous new year together!
Best regards,
ProtoTi Global Business Team


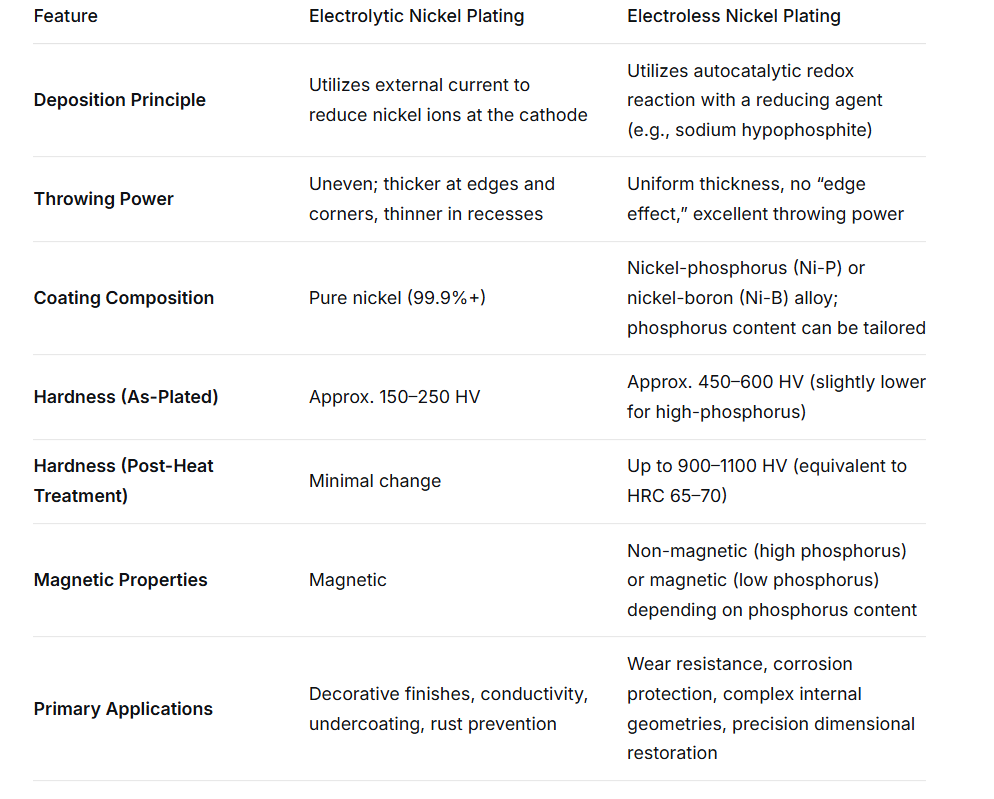
Nickel plating is a surface treatment technology that deposits a layer of nickel coating on the surface of a metal or non-metal substrate through electrochemical or chemical deposition methods. Nickel is a silvery-white, slightly yellowish metal with ferromagnetic properties. It is prone to passivation in the air, forming an extremely thin protective film, thus exhibiting excellent corrosion resistance.
CNC processing typically uses materials such as aluminum, copper, steel, and stainless steel. Each material has its own limitations:
Aluminum/Copper: The texture is relatively soft, with poor wear resistance and prone to oxidation.
Carbon steel: Prone to rust.
Stainless steel: Although it is resistant to corrosion, it is costly and may still corrode in specific environments (such as those containing chlorine).
Applying the nickel plating process to CNC-machined parts can lead to significant performance improvements across multiple dimensions, specifically as follows:
Outstanding corrosion resistance:
Nickel itself has excellent chemical stability and exhibits outstanding corrosion resistance in air, water, alkaline solutions, and various organic acids. The chemical nickel plating layer is dense in structure and has a very low porosity, effectively preventing the contact between the corrosive medium and the base material. Especially for the chemical nickel plating with high phosphorus content (>10% P), due to its amorphous structure, the corrosion resistance is even more outstanding. It can easily pass the 500-hour or even 1000-hour salt spray tests. For CNC processing parts working in marine environments, chemical equipment, or humid conditions, nickel plating is an ideal choice to extend the service life.
Excellent wear resistance and hardness:
The hardness of the freshly deposited electroless nickel coating is typically between 500 and 600 HV, which is already superior to many substrates. More importantly, after heat treatment (usually within the range of 200°C to 400°C), the nickel-phosphorus compounds in the coating will precipitate and disperse for strengthening, significantly increasing the hardness to 850-1100 HV, or even higher. This high hardness characteristic enables nickel-plated parts to withstand high loads and high-frequency friction and wear, and is widely used in hydraulic pistons, mold cavities, bearing housings, and other wear-prone components. Compared to hard chrome, the wear resistance of nickel plating is slightly inferior, but it has better toughness, is less prone to peeling, and has much better uniformity on complex shapes than hard chrome.
Uniform coating thickness:
This is the most significant advantage of chemical nickel plating compared to electroplating. CNC processed parts often have complex features such as threads, deep holes, internal cavities, and sharp corners. The electroplating process is prone to problems like thin coating or inability to cover in these areas. However, chemical nickel plating relies on self-catalytic reactions and can achieve almost uniform coating thickness as long as the solution can come into contact with the surface. This characteristic is crucial for ensuring the dimensional accuracy and fit tolerance of the parts, especially in applications such as precision transmission and sealing fits.
Good weldability and electromagnetic shielding performance:
The nickel plating layer has excellent weldability, especially suitable for electronic components and connectors that require subsequent welding assembly. At the same time, nickel is a ferromagnetic material. The electroless nickel plating layer can provide effective electromagnetic interference (EMI) and radio frequency interference (RFI) shielding, which is of great value in applications such as electronic device casings and communication equipment.
Aesthetic decorative effect:
Although electroless nickel plating is mainly targeted at functional applications, its surface finish is very smooth, presenting a semi-glossy to glossy state, and it also has a certain decorative effect. For parts that require both functionality and an attractive appearance, nickel plating is an ideal solution that takes into account both aspects.
In the field of CNC processing, two types of nickel plating processes are mainly applied: electroplating nickel and electroless nickel plating (without electrolytic nickel).
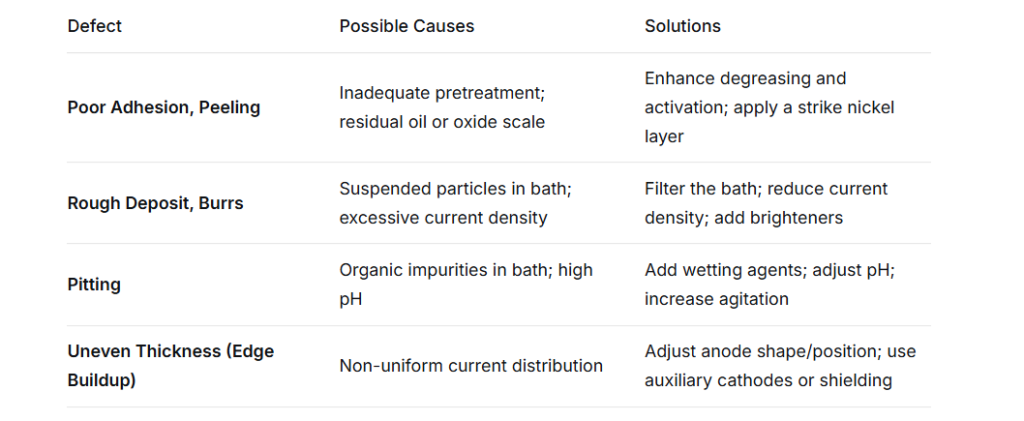
Before the CNC processed parts enter the electroplating line, they must undergo strict pre-treatment, which is the key factor determining the adhesion strength of the coating.
Mechanical polishing / grinding: Remove the tool marks and burrs left by CNC processing, and reduce the surface roughness. This step is crucial for products requiring a mirror finish.
Ultrasonic degreasing: Removes cutting fluids, anti-rust oils and grease from the surface of the parts. Residual oil stains can cause the coating to flake off and fall off.
Pickling activation: Use dilute sulfuric acid or hydrochloric acid to remove the oxide film on the surface, revealing the active metal lattice.
Pre-plating (impact nickel): For materials that are prone to corrosion, such as stainless steel and aluminum alloys, a pre-plating process is carried out first to ensure the adhesion of the base layer.
Electroplating of nickel: In a solution of nickel salts (sulfuric nickel, chlorinated nickel, boric acid), nickel is deposited by applying direct current through a rectifier.
Post-treatment: This includes water washing, passivation (to prevent discoloration), sealing or drying.
Current density: Usually controlled within 2 – 6 A/dm². Excessive current will cause the coating to burn (become rough and black); too low a current will result in a slow deposition rate and loose crystallization.
Temperature: Maintain at 45 – 60℃. Temperature affects internal stress and deposition rate.
pH value: It should be controlled within the range of 3.5 – 4.5. If the pH is too high, it will cause the co-deposition of hydroxide ions, resulting in pinholes; if the pH is too low, the current efficiency will decrease.
Mixing: Air agitation or cathode movement helps to reduce concentration polarization, resulting in a finer coating.
For complex parts such as hydraulic valve blocks, medical implants, and precision gears, electroless nickel plating (EN) is the preferred method.
The greatest advantage of chemical nickel plating lies in its uniform thickness. Since it does not rely on electric current, the deposition rate is basically the same wherever the solution comes into contact. This is incomparable to electroplating for CNC processed parts with complex geometries such as internal threads, deep holes, blind holes, and gear tooth roots.
The phosphorus content in electroless nickel plating determines its physical properties:
Low phosphorus (1-5% P): High hardness, good wear resistance, with certain magnetism, suitable for wear-resistant applications.
Medium phosphorus (5-9% P): It has the best comprehensive performance, combining hardness, wear resistance and corrosion resistance, and is the most widely used.
High phosphorus (10-13% P): Extremely corrosion-resistant (especially in acidic environments), non-magnetic, suitable for electronics, medical and marine engineering.
The chemical nickel plating layer is in an amorphous structure when in the deposited state. Through heat treatment (typically at 300-400℃, with a holding time of 1 hour), the Ni3P phase can be precipitated, increasing the hardness from 500 HV to 900-1000 HV and further enhancing the adhesion. However, it should be noted that heat treatment may cause deformation of the substrate or a decrease in hardness (such as aging softening of aluminum alloys), and the temperature needs to be adjusted according to the characteristics of the substrate.
To better understand the advantages of nickel plating technology, this section will conduct a detailed data comparison to analyze the performance differences between nickel plating and other common surface treatment processes (such as hard chrome, zinc plating, and PVD coating), and explore the optimal matching strategies for it in various application scenarios.
The table below conducts a comprehensive comparison of four mainstream surface treatment processes from multiple aspects:
Performance Comparison Table of Four Surface Treatment Processes
| Performance Indicator | Electroless Nickel Plating (ENP) | Hard Chrome | Zinc Plating | PVD Coating (TiN/CrN, etc.) |
|---|---|---|---|---|
| Coating Hardness (HV) | 500-600 (As-plated) 900-1100 (After Heat Treatment) | 800-1200 | 150-200 | 2000-3500 |
| Wear Resistance | Excellent | Superior | Fair | Exceptional |
| Corrosion Resistance | Excellent (esp. High-P) | Good (requires sealing) | Good (Sacrificial Anode) | Excellent |
| Coating Uniformity | Exceptional (±10%) | Fair (Field-dependent) | Fair | Good (Rotation-dependent) |
| Max Thickness (μm) | 5-100+ | 5-50 | 5-25 | 1-5 |
| Operating Temp (°C) | -200 to +400 | -200 to +600 | <150 | <600 |
| Friction Coefficient | 0.15-0.25 (Dry) | 0.1-0.2 (Dry) | 0.3-0.4 | 0.1-0.4 (Material-dependent) |
| Solderability | Good | Poor | Fair | Poor |
| EMI Shielding | Good | Fair | Fair | None |
| Environmental Impact | Phosphorus wastewater treatment needed | Hexavalent Chromium highly toxic | Relatively Eco-friendly | Very Eco-friendly (Dry Process) |
| Cost | Moderate | Medium-High | Low | High |
| Compatible Substrates | Metals, Some Plastics | Metals | Steel/Iron | Metals, Ceramics |
| Complex Geometry Adaptability | Exceptional | Poor | Moderate | Moderate |
Based on the above performance comparison, we can recommend the most suitable surface treatment solution for different application scenarios:
Preferred option: Hard chromium. Suitable for hydraulic rods, mold cavities, engine cylinder liners, etc., in extremely worn environments.
Alternative option: Chemically plated nickel after heat treatment. When the part shape is complex and hard chromium cannot be evenly coated, chemically plated nickel is the best alternative. It has better toughness than hard chromium and is less prone to peeling.
Preferred: High phosphorus electroless nickel plating (>10% P). Its amorphous structure provides unparalleled corrosion resistance, suitable for marine platform components, chemical valves, ship fittings, etc.
Alternative: Galvanization (thick coating) + sealing treatment. Lower cost, suitable for general outdoor steel structures, but has a shorter lifespan than nickel plating in chloride-containing environments.
The only choice: Chemical nickel plating. Its outstanding uniform plating ability ensures the dimensional consistency of complex features such as threads, deep holes, and internal cavities. It is the ideal choice for precision instruments, optical equipment, and semiconductor equipment parts.
Preferred options: Hard chromium or PVD coating. Hard chromium can withstand temperatures up to 600°C, and PVD coatings (such as CrN) also have excellent high-temperature stability.
Limitations: The long-term use temperature of chemical nickel plating should not exceed 400°C; otherwise, the coating may oxidize or soften.
Preferred option: Low-phosphorus electroless nickel plating or PVD coating. This avoids the toxicity of hexavalent chromium and complies with strict regulations such as those of FDA and EU.
Trend: As environmental protection regulations become increasingly strict, the use of hexavalent chromium hard chrome is being subject to more and more restrictions. Trivalent chromium electroplating and PVD technology are rapidly replacing traditional hard chrome.
Although the one-time processing cost of electroless nickel plating is higher than that of zinc plating, considering the performance improvement and extended lifespan it brings, the life cycle cost (LCC) of electroless nickel plating often has a more favorable advantage. For example, in the application of the impeller of a chemical pump, the zinc-plated impeller may need to be replaced every six months, while the nickel-plated impeller can be used for 3 to 5 years. When calculated comprehensively, the total cost of the nickel-plating solution is lower, and it also reduces the downtime for maintenance.

Components: High-pressure Common Rail Tube, Injector Body.
Base Material: Alloy Steel.
Process: Chemical Nickel Plating (Medium Phosphorus).
Reason: The internal channels are complex, requiring uniform coating thickness; it needs to withstand a fuel pressure of up to 2000 bar and corrosive (sulfur-containing) fuel; The chemical nickel coating provides excellent pressure resistance sealing and corrosion resistance.
Part: Hydraulic actuator.
Base material: 300M ultra-high strength steel.
Process: Electroplating cadmium (traditional) or chemical nickel plating (alternative).
Reason: To meet strict hydrogen embrittlement control requirements; chemical nickel plating replaces the toxic cadmium plating process, providing dual protection of wear resistance and corrosion prevention.
Part: Aluminum alloy body frame.
Base material: 6063 aluminum alloy.
Process: Bright nickel electroplating (under layer) + Gold plating/Chrome plating (surface layer).
Reason: Provide a high-gloss decorative appearance, and as a conductive layer, meet the requirements for antenna signal transmission. The high-precision shape processed by CNC still maintains dimensional tolerance after plating.
Parts: Orthopedic drills, scissors.
Base material: Stainless steel or titanium alloy.
Process: Chemical nickel plating (high phosphorus).
Reason: Required to be non-magnetic (to avoid interference with MRI), biocompatible, excellent wear resistance (resistant to repeated disinfection).
The increasingly stringent environmental regulations such as the EU REACH regulations and China’s “Emission Standards for Electroplating Pollutants” are driving the nickel plating industry towards a more environmentally friendly direction.
Chromium-free passivation: The traditional chromate passivation is being gradually phased out due to the extremely toxic nature of hexavalent chromium, and is being replaced by chromium-free passivation technologies such as molybdate, tungstate, and silane.
Wastewater treatment upgrade: For the nickel ions and phosphites contained in the chemical nickel plating wastewater, new technologies such as chemical precipitation, ion exchange, and membrane separation are being widely applied to achieve compliance discharge and even zero discharge.
Long-life plating solution: Developing more stable and longer-lasting plating solutions to reduce the amount of waste liquid.
Single-function nickel plating layers have struggled to meet the diverse demands of high-end applications. Composite coatings have thus become a research hotspot.
Ni-P-SiC, Ni-P-Diamond: By introducing hard particles such as silicon carbide and diamond into the plating solution, composite coatings are formed, further enhancing wear resistance and suitable for extreme wear conditions.
Ni-P-PTFE: Introducing PTFE (polytetrafluoroethylene) particles significantly reduces the friction coefficient and achieves self-lubrication, suitable for oil-free lubrication environments.
Nanomodified coatings: Utilizing nanotechnology to refine the crystal grains of the coating, improving density and overall performance.
Under the Industrial 4.0 wave, the nickel plating production line is gradually undergoing an intelligent upgrade.
Online Monitoring: Sensors are used to monitor parameters such as the temperature, pH value, and component concentration of the plating solution in real time, and automatically adjust to ensure process stability.
Big Data Analysis: Collecting massive data from the production process, using AI algorithms to analyze the causes of defects, predict equipment failures, and optimize process parameters.
Automated Loading and Unloading: Robots replace manual operations for parts loading and unloading, improving production efficiency and reducing human errors.
With the widespread adoption of metal 3D printing technology, the high surface roughness and complex internal structure of its parts have presented new challenges for surface treatment. Chemical nickel plating, due to its excellent uniform plating ability and filling capacity, has become an ideal choice for post-processing of 3D printed parts, effectively improving surface quality and enhancing mechanical properties.

Nickel plating technology, as an indispensable part of the CNC machining industry chain, is becoming increasingly important. From the in-depth understanding of the process principle, to the meticulous planning in the design stage, and to the strict control in the production process, every step is crucial to the success or failure of the final product. Facing increasingly fierce market competition and strict environmental requirements, manufacturing enterprises can only innovate continuously, embrace new technologies, optimize process flows, and thus stand unassailable in the high-end manufacturing field.
This article aims to provide practitioners with a comprehensive and practical technical guide, hoping to help enterprises achieve greater breakthroughs in nickel plating application. In the future, with the cross-disciplinary integration of materials science, chemical engineering, and intelligent manufacturing, nickel plating technology will surely regain new vitality and contribute more to the progress of human society.

Start your project today. Get free DFM from professional engineer at ProtoTi.
Share the Post:

Additive manufacturing has established itself as a fundamental technology in rocket development, especially in the production of combustion chambers, as it allows

SLA (Stereolithography) is a 3D printing technology that uses a UV laser to cure liquid photopolymer resin layer by layer,

BJ (Binder Jetting) is becoming a popular choice for mass-producing metal parts, with its biggest highlight being: no support structures needed at all.

In the grand narrative of modern industrial civilization, every minor advancement in materials science often leads to a significant leap in human productivity. Among various metal materials, stainless steel undoubtedly ranks as one of the most revolutionary inventions. It not only transformed the appearance of buildings, redefined safety standards in healthcare, but also became the cornerstone in key fields such as chemical engineering, marine, and energy. Within the vast family of stainless steel, 316 stainless steel (and its low-carbon variant 316L) is hailed as the “industrial gold” due to its outstanding comprehensive performance, and is the preferred material in highly corrosive environments.